(a) Name the device used for producing an electric current from a chemical
(b) Copy and complete the table below.
|
Electrolyte |
Product at the anode (carbon) |
Product at the cathode (carbon) |
|
Dilute NaCl\(_{(aq)}\) Concentrated NaCI\(_{(aq)}\) |
(a) Mention the chemical substance manufactured starting from each of the foirownc sets of materials:
(i) sugar and yeast;
(ii) ammonia, air and water;
(iii) vegetable oil and caustic alkali.
(b) State one air pollutant generated during the manufacture of fertilizers.
(a) What is the change in oxidation state of chromium in the reaction represented by the following equation?
3SO\(_2\) + Cr\(_2\)O\(^2_{-7}\) + 2H\(^+\) -> 3SO\(_4^{2-}\) + 2Cr\(^{3+}\) + H\(_2\)O
(b) Use the half equations given below to deduce the equation for the reaction between iron(II) ions and heptaoxodichromate (VI) ions in acidic solution.
Fe\(^{2+}\) --> Fe\(^{3+}\) + e\(^-\)
Cr\(_2\)O\(^{2-}_7\) + 14H\(^+\) + 6e\(^-\) ----> 2Cr\(^{3+}\) + 7H\(_2\)O.
(a) Mention one process apart from respiration, which increases the amount of carbon (IV) oxide in the atmosphere.
(b)(i) State one use of sodium hydrogentrioxocarbonate (IV).
(ii) Write an equation to show the action of heat on sodium hydrogentrioxocarbonate(IV).
(a) List two differences between solids and liquids.
(b) The graph below is the heating curve for a solid X. Use the graph to answer Questions (i) — (iii) below.
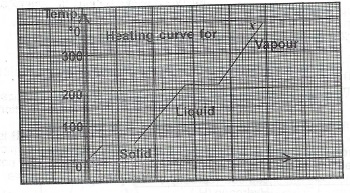
(i) What is the melting point of X?
(ii) If the vapour of X is cooled, at what temperature will it start to condense?
(iii) (I) As X is heated, state what happens to the: I. frequency of collision of molecules of X;
(II) value of the entropy of the system.