(1)(a) State Avogadro's law
(b) Which of the state of matter contains particles that are: (i) readily used
(ii) held firmly together by some forces of cohesion;
(iii) involved in rapid random motion?
A shortened form of the Periodic Table is shown below. Use it to answer questions (a) and (b)
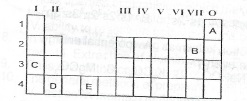
(a) Which of the elements represented as A to E in the table, above is:
(i) transition metal;
(ii) an alkaline earth meta
(iii) the least reactive;
(iv) the most electronegative?
(b)(i) What type of bond would exist in a compound formed when element D reacts with oxygen?
(ii) Write the formula of the compound formed in (b)(i) above.
(a) Give one example each of a naturally occurring substance that;
(i) conforms to the general formula C(_x\)(H\(_2\)O)\(_y\);
(ii) contains the carboxyl group as its functional group.
(b) Name the process for obtaining: (i) paraffin oil from crude oil; (ii) benzene from ethyne.
(a) Define oxidation in terms of electron transfer.
(b) Consider the following: Cu\(_{(s)}\) + 2Ag\(^{+}_{(Ag)}\) \(\to\) Cu\(^{2+}_{(aq)}\) + 2Ag\(_{(s)}\)
(i) State the species that is reduced
(ii) Write half-cell equation for each of the species.
(a) When MgSO\(_4\).XH\(_2\)O crystals were exposed to the atmosphere for several days, three was a loss in mass
(i) What name is given to this phenomenon?
(ii) Give another example of a compound that exhibits this phenomenon
(b) If 0.50 mole of MgSO\(_4\).XH\(_2\)O has a mass of 123g. calculate the value of X. (H = 1, O = 16, Mg = 24, S = 32)