(a) The elements listed below to the same group in the Periodic Table \(_9F\) \(_{17}CI\) \(_{35}Br\) \(_{53}I\).
(i) Write the electronic structure of the first member
(ii) What is the family name of the elements.
(iii) Which of the elements has the strongest oxidizing ability?
(b) Use the information provided in the following table to answer Questions (i) to (ii) below
(b)(i) Which of the chlorides would exist as discrete molecules?
(ii) What type of bonding holds atoms of A and chlorine together in ACI?
(iii) Which of the chlorides would be a good conductor of electricity?
(a) (i)Name the class of oxides to which copper (II) oxide belongs.
(ii) Give the reason why copper (II) oxide increases in mass on exposure to the atmosphere
(iii) Write an equation to show the action of dilute hydrochloric acid on copper (II) oxide.
(b) List two methods of removing total hardness from water.
Use the following energy profile diagram to answer Questions (a) to (c) below.
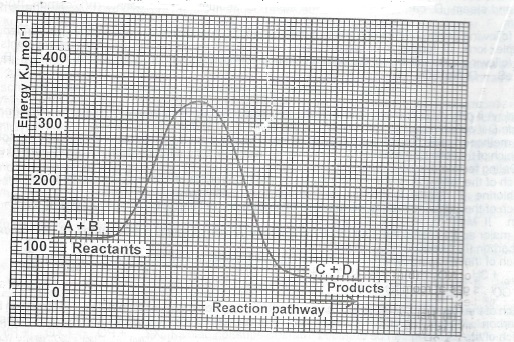
(a) From the diagram, determine the value of the; (i) enthalpy of the reaction (ii) activation energy of the reaction.
(b) State whether the profile diagram is for an endothermic reaction or an exothermic reaction.
(c) What name is given to substances which can provide an alternative pathway for the reaction?
(a) Give the name of the:
(i) process for obtaining ethanol from sugars;
(ii) compound produced when ethanol undergoes bacterial oxidation.
(iii) enzyme in yeast which catalyses the conversion of maltose to glucose.
(b)(i) Write the structural formula of ethanol
(ii) Name the organic product formed when propanoic acid reacts with ethanol.
(a) State the monomer units of (i) polyethene; (ii) cellulose
(b) Name the two fuels obtained when steam and air are passed over red-hot coke.