(a)i. With the aid of an equation, explain briefly why aluminum metal is not affected by air.
(ii) In the extraction of aluminum from bauxite, state the:
- substance used for purifying the ore;
- composition of the mixture electrolyzed.
(b) ZnO is an amphoteric oxide. Write equations to illustrate this statement.
(c)i) List three uses of sodium trioxocarbonate(IV).
(ii) Explain briefly why a solution of trioxonitrate(V) acid turns yellowish on storage for some time.
(ii) Describe briefly how trioxonitrate(V) ions could be tested for in the laboratory.
(d) Write balanced chemical equations for the preparation of hydrogen chloride.
(i) using concentrated H\(_{2}\)SO\(_{4}\):
(ii) by direct combination of its constituent elements.
(iii) State one use of hydrogen chloride.
(a)(i) Define the first ionization energy of an element
(ii) Consider the following table and use it to answer te question that follows
| Element | Li | Be | b | C | N | O | F | Ne |
| Atomic number | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 |
| 1st I.E/kj mol\(^{-1}\) | 520 | 900 | 801 | 1086 | 1402 | 1314 | 1681 | 2081 |
Explain briefly why the first ionization energy of B is less than that of Be despite the fact that the atomic number of B is greater than that of Be.
(b) When Titanium chloride was electrolysed by passing 0.12 A current through the solution for 500 seconds, 0.015 g of titanium was deposited. What is the charge on the titanium ion?
[ IF= 96500 C, Ti= 48.0 ]
(c)(i) Aluminium can be obtained by the application of electrolysis. State the electrolyte which yields aluminium on electrolysis.
(ii) Name two major factors which would favour the siting of an aluminium smelter in a country.
(d)(i) Define the term paramagnetism.
(ii) Consider the following ions: 24\(^{Cr ^{2+}}\), 24\(^{Cr^{6+}}\)
(I) Deduce the number of unpaired electrons in each of the ions.
(II) State which of the ions will have a greater power of paramagnetism
(l) Give a reason for the answer stated in (d)(ii)(II)
(a) (i) Define the term Avogadro's number.
(ii) If 2.30 g of an oxide of nitrogen, x, contains 3.01 x 10\(^{22}\) molecules, calculate the molar mass of x.
(iii) Deduce the formula of x. N, =6.02 x 10", N =14.0, O = 16.0]
(b)(i) Describe briefly what happens when each of the following substances are added to water:
(I) CCI\(_{4}\); (II) SiCI\(_{4}\),
(ii) Explain briefly why the reactions in (a)(i), (b)(i), (I) and (b)(ii) (II) are different Study the diagram below and answer the questions that follow.
(c) Study the diagram below and answer the questions that follow.

(i) What is the set up used for?
The sub-atomic particles located in the nucleus of an atom are?
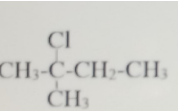
The IUPAC nomenclature of the structure is?