An open-tube mercury manometer is used to measure the pressure in a gas tank. When the atmospheric pressure is 101,325 \(P_a\), what is the absolute pressure in \(P_a\) in the tank if the height of the mercury in the open tube is 25 cm higher. density of mercury = \(13600kg/m^3, g = 9.8m/s^2\)
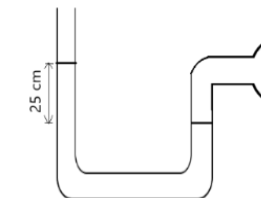
density (ρ) = 13600 \(kgm^{-3}\); g =\(9.8 ms^{-2}\)
\(P_{atm}\) =101,325 Pa; ρ=13600 \(kgm^{-3}\)
Pabs=\(P_{atm}\)+ρgh
⇒Pabs=101,325+13600 x 9.8 x 0.25
⇒Pabs=101,325+33320
⇒ Pabs=134,645Pa
There is an explanation video available below.
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}