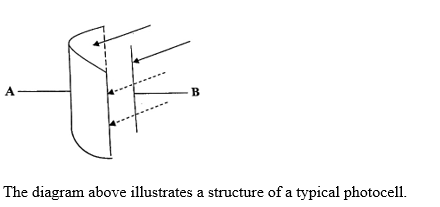
The diagram above illustrates a structure of a typical photocell.
(i) Identify each of the parts labelled A and B.
(ii) State one function each of A and B
(iii) Einstein’s photoelectric equation can be written as E = hf – Wo. State what each of the terms E, hf and Wo represent.
(b) A photon is incident on a metal whose work function is 1.32 eV. An electron is emitted from the surface with a maximum kinetic energy of 1.97 eV. Calculate the frequency of the photon. [1 eV = 1.6 x 10-19 J]
(c)(i) Define half-life of a radioactive element.
(ii) Sketch a graph of the relation N = Noe-λt and indicate the half-life.
The expected answer is:
(a) (i) Identification of labelled parts of photocell
A - Emitter / cathode
B - Collector / anode
(ii) Function of part of photocell
A - emits electrons
B - attracts electrons
(iii) Meaning of terms in Einstein’s photoelectric equation
E - kinetic energy of photoelectrons
hf - energy of incident photon
Wo - work function of metal

Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}