(a) Define the boiling it of a liquid.
(b) Describe with the a d. labelled diagram, an experiment to determire the boiling point of a small quantity of a liquid.
(c) factors that may affect the boiling point of a liquid
(d) Using the kinetic theory of matter, explain why pure water changes to steam at S.T.P. without any change in temperature, although heat is being supplied to the water.
a) The boiling pointer of a liquid is the temperature at which its s.v.p. is equal to the external atmospheric pressure OR The temperature at which a (pure) liquid boils when the atmospheric pressure on it is 760 mm Hg. OR The temperature at which the liquid changes from the liquid to the gaseous state without change in temperature when the atmospheric pressure is normal (760mm Hg).
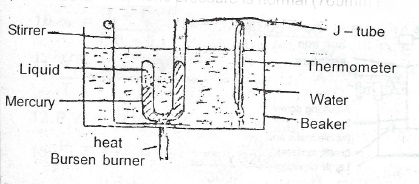
Method: Some mercury is introduced into J - tube and the entrapped air removed. Some liquid is introduced into the shorter length of the J - tube above the mercury. The beaker and its contents are heated and the water stirred continuously. At a stage when the levels of mercury in the two limbs of the J-tube are the same the temperature is read. At this temperature the s.v.p of the liquid is equal to the atmospheric pressure, and this temperature is the boiling point of the liquid. The process is repeated with the beaker and its contents cooling. The temperature at which the levels of mercury in the two limbs are equal is read and the mean of the two taken.
(c) Pressure on the liquid, impurities in the liquid.
(d) The extra heat supplied during boiling is used in breaking the intermolecular attractions and in mabling the steam expand or enabling the molecules move further apart against the atmospheric pressure. Hence, there is no change in temperature during the change of state.
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}