(a) Explain the following terms:
(i) mass defect;
(ii) binding energy of a nucleus.
(b)(i) Assuming the wave nature of an electron, what is the effect of decreasing the speed of a photoelectron on its; (\(\alpha\)) wavelength? (\(\beta\)) energy?
(ii) A particle of friasS 4.4 x 10\(^{-23}\) kg moves with a velocity of 10\(^5\)ms\(^{-1}\). Calculate its wavelength. (h = 6.6 x 10\(^{-34}\) Js)
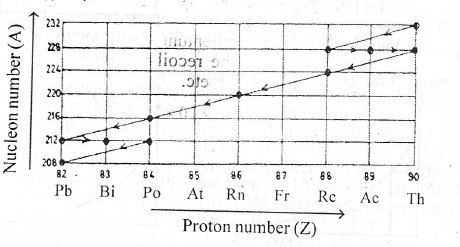
The diagram above shows part of a radioactive decay series. Use it to answer the following questions.
(i) Name a pair of isotopes.
(ii) Name the isotopes with which the series starts.
(iii) Write down a nuclear equation for two ekgmples of each of: (\(\alpha\)) alpha decay; (\(\beta\)) beta decay.
(a)(i) Mass defect is the difference between the mass of a nucleus and the total mass of its nucleons. It expressed in atomic mass unit (a.m.u.). It is also a measure of binding energy.
Mass defect = total mass of nucleus
– mass of nucleons.
(ii) Binding energy is the work done or energy needed to separate the nucleons in a nucleus. It is measured in eV or Joules (J).
(b)(i) The effect of decreasing the speed of a photoelectron on: (\(\alpha\)) wavelength --> wavelength increases. (\(\beta\)) energy ---> energy decreases.
(ii) \(\lambda = \frac{h}{mv}\)
= \(\frac{6.6 \times 10^{-34}}{4.4 \times 10^{-2} \times 10^{-16}}\)m
(c) Pairs of isotopes in the figure are;
\(^{208}_{82}Pb\) and \(^{212}_{82}Pb\) or \(^{212}_{84}Po\) and \(^{216}_{84}Po\) or
\(^{224}_{88}Ra\) and \(^{288}_{88}Ra\) or \(^{288}_{90}Th\) and \(^{232}_{90}Th\)
(ii) The series started with isotopes \(^{232}_{90}Th\)
(iii) Nuclear equation for;
\(\alpha\)Alpha decay
\(^{232}_{90}Th\) \(\to\) \(^{228}_{88}Ra\) + \(^{4}_{2}He\) + Energy;
\(^{228}_{90}Th\) \(\to\) \(^{22}_{88}Ra\) + \(^{4}_{2}He\) + Energy;
\(^{224}_{88}Ra\) \(\to\) \(^{220}_{86}Rn\) + \(^{4}_{2}He\) + Energy;
\(^{212}_{84}Po\) \(\to\) \(^{208}_{82}Pb\) + \(^{4}_{2}He\) + Energy;
\(^{216}_{84}Po\) \(\to\) \(^{212}_{82}Pb\) + \(^{4}_{2}He\) + Energy;
\(^{212}_{84}Rn\) \(\to\) \(^{208}_{82}Po\) + \(^{4}_{2}He\) + Energy;
\(^{220}_{84}Rn\) \(\to\) \(^{216}_{84}Po\) + \(^{4}_{2}He\) + Energy;
\(^{216}_{84}Rn\) \(\to\) \(^{212}_{82}Pb\) + \(^{4}_{2}He\) + Energy;
(\(\beta\)) Beta decay
\(^{224}_{88}Ra\) \(\to\) \(^{224}_{89}Ac\) + \(^{o}_{-1}e\) + Energy;
\(^{228}_{88}Ac\) \(\to\) \(^{228}_{90}Ac\) + \(^{o}_{-1}e\) + Energy;
\(^{212}_{82}Pb\) \(\to\) \(^{212}_{83}Bi\) + \(^{o}_{-1}e\) + Energy;
\(^{212}_{83}Ra\) \(\to\) \(^{212}_{84}Ac\) + \(^{o}_{-1}e\) + Energy;
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}