(a) Define stable equilibrium as applied to a rigid body.
(b) Sketch a block and tackle system of pulleys with a velocity ratio of 3.
(c) At the beginning of a race, a tyre of volume 8.0 x 10\(^{-4}\) at 20°C-has a gas pressure of 4.5 x 10\(^5\) Pa. Calculate the temperature of the gas in the tyre at the end of the race if the pressure has risen to 4.6 x 1 0\(^5\) Pa.
(d)(i)
|
Ice point 273k |
373k Steam point |
|
|
Resistance/\(\Omega\) |
5.67 | 7.75 |
|
Pressure/Pa |
7.13 x 10\(^4\) | 9.74 x 10\(^4\) |
The table above shows readings of the resistance and pressure of a platinum resistance thermometer and a constant-volume gas thermometer respectively, when immersed in the same liquid bath. Use this data to determine the temperature of the bath on the: (\(\alpha\)) resistance thermometer; (\(\beta\)) gas thermometer
(ii) By what percentage is the temperature measured on the platinum resistance thermometer in error?
(a) A body is said to be in stable equillibrium if it tends to return to its original position after being slightly displaced.
(b)
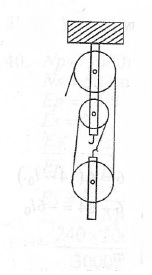
i.e. V.R = Number of pulleys in the system
V.R = 3
(c) \(P_1 = 4.5 \times 10^5\)Pa; \(_2 = 4.6 \times 10^5\)Pa
T\(_1\) = 20\(^{o}\) + 273 = 293k
T\(_2\) = ?
\(\frac{P_1}{T_1} = \frac{P_2}{T_2}; \frac{4.5 \times 10^5}{293} = \frac{4.6 \times 10^5}{T_2}\)
\(T_2 = \frac{4.6 \times 10^5 \times 293}{4.5 \times 10^5}\)
\(T_2\) = 299.5k or 26.5\(^o\)C
(d) \(\alpha\) Resistance thermometer
\(\frac{a}{b} = \frac{c}{d}; \frac{R \theta - 5.67}{7.75 - 5.67} = \frac{R - 273}{373 - 273}\)
\(\frac{R \theta - 5.67}{1,88} = \frac{R - 273}{100}\)
\(\beta\) Gas thermometer
\(\frac{a}{b} = \frac{c}{d}; \frac{G\theta - (7.13) x 10^4}{(9.74 - 7.13) \times 10^4} = \frac{G - 273}{373 - 273}\)
\(\frac{G\theta - 7.13}{2.16} = \frac{G - 273}{100}\)
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}