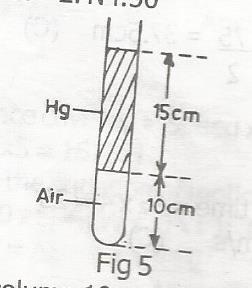
An air column 10cm in length is trapped into the sea end of a capillary tube by a 15cm column of mercury with the tube held vertically as shown above. On inverting the tube the air column becomes 15cm long. The atmospheric pressure during the experiment is
Let X represent the atmospheric pressure
with tube upright,P1 = ( x + 15)cm, L1 = 10cm
with tube inverted P2 = ( x - 15)cm, L2 = 15cm
using Boyle's law i.e P1V1 = P2V2
(x + 15)10 = (x - 15)15 = 10x + 150 = 15x - 225
15x - 10x = 150 + 225
5x = 375
x = \(\frac{375}{5}\)
= 75cm
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}