Physics
JAMB 1999
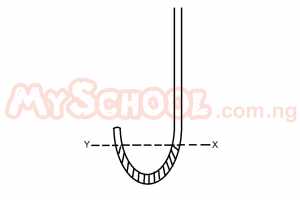
In the J-tube above, Y and X are on the same horizontal level and 30cm
3 of air is trapped above Y when the atmospheric pressure is 75cm Hg, calculate the volume of air trapped above Y when 15cm Hg is now poured into the limb above X
-
A.
15 cm3
-
B.
25 cm3
-
C.
35 cm3
-
D.
45 cm3
Correct Answer: Option B
Explanation
Since the temperature of the air is to remain Constant => P
1V
1 = P
2V
2
∴ 75 x 30 = (75 + 15) x V
2
75 x 30 = 90 x V
2
= 25cm
2
Report an Error
Ask A Question
Download App
Quick Questions
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}