(a) (i) State three characteristic properties of transition metals.
(ii) What is the oxidation state of manganese In each of the following species? (1) MnCl\(_2\) (II) MnO\(_2\) (III) MnO\(_4^{-}\)
(iii) Explain why manganese conducts electricity in the solid state but manganese chloride conducts only when molten or in solution.
(b)(i) The collision theory suggests that for two particles to react, they must collide. What two factors determine whether or not the collision would lead to formation of products?
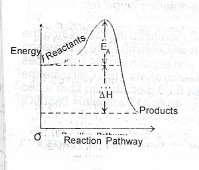
(ii) Use an energy profile diagram to illustrate what is meant by the enthalpy change (\(\Delta\)H) and the activation energy (E\(_A\)) of a reaction.
(c) When few drops of aqueous KSCN are added to a solution of iron (III) salt the following equilibrium is set up:
Fe\(^{3}_{(aq)}\) + 3SCN\(^{-}_{(aq)}\) \(\rightleftharpoons\) Fe(SCN)\(_{3(aq)}\)
yellow colourless deep red
The equilibrium mixture has a pale red colour.
(i) Explain what would happen if more KSCN\(_{(aq)}\) were added to the equilibrium mixture.
(ii) Which of the ions in the equilibrium mixture forms an insoluble hydroxide with NaOH\(_{(aq)}\)? Write an equation for the reaction
(iii) State two changes observed on adding NaOH\(_{(aq)}\) to the equilibrium mixture.
(a)(i) Characteristic properties of transition metals.
– Variable oxidation states
– Complex ion. formation
– Formation of coloured ions/compounds
– Paramagnetism – Catalytic ability.
(ii) The oxidation state of manganese in I. MnCl\(_2\) is +2 II. MnO\(_2\) is +4 III. MnO\(^-_4\) is +7
(iii) Manganese conducts electricity in the solid state because it consists of metal nuclei and a sea of mobile (valence) electrons which conduct the current. Manganese chloride conducts only when molten or solution because conduction is by mobile ions.
(b)(i) Factors that determine whether or not the collision of reactant particles would result in the formation of products
–Energy of collision/colliding particles (temperature or activation energy)
–Alignment of the particles.
(ii)
(c) Fe\(^{3+}_{(aq)}\) + 3SCN\(^-_{(aq)}\) \(\rightleftharpoons\) (SCN)\(_{(aq)}\)
yellow colourless deep red
Equilibrium mixture has a pale red colour
(i) On adding more of KSCN\(_{(aq)}\) to the equilibrium mixture, the red colour will deepen. Addition of KSCN would increase the concentration of SCN\(^-\) causing equilibrium to shift to Products the right.
(ii) Fe\(^{3+}\) reacts with NaOH ; Fe\(^{3+}_{(aq)}\) + 3OH\(^-\) --> Fe(OH)\(_3\).
(iii) On adding NaOH\(_{(aq)}\) to the equilibrium mixture, a brown precipitate is formed, a colourless solution is obtained/the red colourpale disappears.
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}