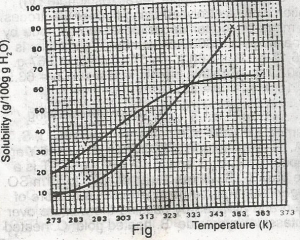
The diagram shown above represents the solubility curves of two salts, X and Y, in water, use this diagram to answer the question. If the molar mass of X is 36 g, the number of moles of X dissolved at 343 K is
The molar mass of x = 36g. At 343k, 72g of x dissolved
Molarity of x = \(\frac{72}{36}\) = 2.00moles.
Contributions ({{ comment_count }})
Please wait...
Modal title
Report
Block User
{{ feedback_modal_data.title }}